Diagram of ion exchange chromatography
Home » Background » Diagram of ion exchange chromatographyYour Diagram of ion exchange chromatography images are ready. Diagram of ion exchange chromatography are a topic that is being searched for and liked by netizens now. You can Download the Diagram of ion exchange chromatography files here. Download all royalty-free photos and vectors.
If you’re searching for diagram of ion exchange chromatography pictures information linked to the diagram of ion exchange chromatography interest, you have visit the right site. Our website frequently provides you with suggestions for viewing the maximum quality video and picture content, please kindly search and locate more informative video articles and graphics that fit your interests.
Diagram Of Ion Exchange Chromatography. The eluent is delivered to the system using a high-pressure pump. The charged molecules in the sample are separated by the electrostatic forces of attraction when they pass through the ionic resin at particular pH and temperature. Some of the charged regions which will influence ion exchange Interaction between opposite charges Charged groups on the proteins interact with charged groups on the ion exchanger. Ion exchange due to competitive ionic binding attraction and ion exclusion due to repulsion between similarly charged analyte ions and the ions fixed on the chromatographic support play a role in the separation in ion chromatography.
 Ion Exchange Chromatography Theory Principle Factors Affecting Prot Ion Exchange Theories Functional Group From pinterest.com
Ion Exchange Chromatography Theory Principle Factors Affecting Prot Ion Exchange Theories Functional Group From pinterest.com
Water is made of water molecules formula H2O. It is widely used as a preparative technique for Atomic Absorption Spectroscopy AAS Multiple Collector Inductively Coupled Plasma Mass. Schematic diagram of an anion exchange media particle. Different amino acids or other substances are adsorbed on the column are then eluted with suitable solvents. Schematic Diagram of Ion exchange chromatography Column. The charged molecules in the sample are separated by the electrostatic forces of attraction when they pass through the ionic resin at particular pH and temperature.
These methods are ion pair chromatography 3 ion exchange chromatography 4 and ion exclusion chromatography 5Analyte and mobile phase are initially always polar andor ionic.
It is widely used as a preparative technique for Atomic Absorption Spectroscopy AAS Multiple Collector Inductively Coupled Plasma Mass. At a pH equal to a proteins pI the protein carries no net charge. The sample is introduced then flows through the guard and into the analytical ion-exchange columns where the ion-exchange. Different proteins have different charges and interact differently. The high efficiency of RPC columns compared with columns used in ion exchange or ion chromatography also makes IPC a valuable alternative to these techniques. These methods are ion pair chromatography 3 ion exchange chromatography 4 and ion exclusion chromatography 5Analyte and mobile phase are initially always polar andor ionic.
 Source: pinterest.com
Source: pinterest.com
By varying the mobile phase concentration of the ion pair reagent. The sample is introduced then flows through the guard and into the analytical ion-exchange columns where the ion-exchange. A typical ion chromatography consists of several components as shown in Figure 3. Anion Exchange Chromatography Principles. Ion Exchange Chromatography IEC or IEX is driven by interactions between charged target molecules and charged immobilized ligands on the chromatography resin.
 Source: pinterest.com
Source: pinterest.com
Ion Exchange chromatography Principle. Ion exchange starts with the equilibration of the exchanger using pH and ionic strength. All natural waters contain some foreign substances usually in small amounts. The sample is introduced then flows through the guard and into the analytical ion-exchange columns where the ion-exchange. Salt Gradients As in most other modes of chromatography SEC being the exception a protein sample is injected onto the column under conditions where it will be strongly retained.
 Source: pinterest.com
Source: pinterest.com
Ion chromatography includes all chromatographic methods that separate ionic substances and substances that dissociate easily. The pI of most proteins is in the range of 5-9. Two distinct mechanisms as follows. A proteins net surface charge changes with pH in a manner that is dictated by a proteins isoelectric point or pI. Matrix has an ion load opposite to that of the protein to be separated and the affinity of the protein to the column is.
 Source: pinterest.com
Source: pinterest.com
Anion Exchange Chromatography Principles. A typical ion chromatography consists of several components as shown in Figure 3. Ion exchange due to competitive ionic binding attraction and ion exclusion due to repulsion between similarly charged analyte ions and the ions fixed on the chromatographic support play a role in the separation in ion chromatography. Different amino acids or other substances are adsorbed on the column are then eluted with suitable solvents. Ion exchange chromatography is the reversible adsorption of charged molecules to immobilized ion groups on a matrix of an opposite charge.
 Source: pinterest.com
Source: pinterest.com
Ion Exchange chromatography Principle. In column chromatography or ion-exchange chromatography various solid adsorbents like starch cellulose or an ion- pig. The sample is introduced then flows through the guard and into the analytical ion-exchange columns where the ion-exchange. Ion exchange starts with the equilibration of the exchanger using pH and ionic strength. Ion exchange chromatography represents a versatile analytical technique that separates ions and polar molecules in a solution mobile phase based on their affinities for an ion exchanger stationary phase.
 Source: pinterest.com
Source: pinterest.com
The high efficiency of RPC columns compared with columns used in ion exchange or ion chromatography also makes IPC a valuable alternative to these techniques. 24 exchange resins are placed as columns in glass tubes Fig. Introduction In ion exchange chromatography retention is based on the attraction between the solute ions and charged sites bound to stationary phase Columns used for ion exchange are characterized by the presence of charged groups covalently attached to the stationary phase Anion exchangers contain bound positive groups where as cation. The separation occurs by reversible exchange of ions between the ions present in the solution and those present in the ion exchange resin. A typical ion chromatography consists of several components as shown in Figure 3.
 Source: pinterest.com
Source: pinterest.com
Cation exchange chromatography in which positively charged species in the mobile phase bind to a negatively charged ligand on the resin. Introduction In ion exchange chromatography retention is based on the attraction between the solute ions and charged sites bound to stationary phase Columns used for ion exchange are characterized by the presence of charged groups covalently attached to the stationary phase Anion exchangers contain bound positive groups where as cation. It is widely used as a preparative technique for Atomic Absorption Spectroscopy AAS Multiple Collector Inductively Coupled Plasma Mass. Anion Exchange Chromatography Principles. A schematic diagram of overall ion exchange chromatography instrumentation is given below the picture Inert materials like Kel-F Teflon and polypropylene are used for the construction of columns in ion exchange chromatography procedure.
 Source: pinterest.com
Source: pinterest.com
Two distinct mechanisms as follows. At a pH equal to a proteins pI the protein carries no net charge. The eluent is delivered to the system using a high-pressure pump. It is widely used as a preparative technique for Atomic Absorption Spectroscopy AAS Multiple Collector Inductively Coupled Plasma Mass. Ion exchange due to competitive ionic binding attraction and ion exclusion due to repulsion between similarly charged analyte ions and the ions fixed on the chromatographic support play a role in the separation in ion chromatography.
 Source: pinterest.com
Source: pinterest.com
Cation exchange chromatography in which positively charged species in the mobile phase bind to a negatively charged ligand on the resin. Some of the charged regions which will influence ion exchange Interaction between opposite charges Charged groups on the proteins interact with charged groups on the ion exchanger. The separation occurs by reversible exchange of ions between the ions present in the solution and those present in the ion exchange resin. The high efficiency of RPC columns compared with columns used in ion exchange or ion chromatography also makes IPC a valuable alternative to these techniques. Ion exchange chromatography represents a versatile analytical technique that separates ions and polar molecules in a solution mobile phase based on their affinities for an ion exchanger stationary phase.
 Source: pinterest.com
Source: pinterest.com
Introduction In ion exchange chromatography retention is based on the attraction between the solute ions and charged sites bound to stationary phase Columns used for ion exchange are characterized by the presence of charged groups covalently attached to the stationary phase Anion exchangers contain bound positive groups where as cation. The high efficiency of RPC columns compared with columns used in ion exchange or ion chromatography also makes IPC a valuable alternative to these techniques. The pI of most proteins is in the range of 5-9. Different amino acids or other substances are adsorbed on the column are then eluted with suitable solvents. Different proteins have different charges and interact differently.
 Source: pinterest.com
Source: pinterest.com
Salt Gradients As in most other modes of chromatography SEC being the exception a protein sample is injected onto the column under conditions where it will be strongly retained. By varying the mobile phase concentration of the ion pair reagent. Charge and exchangeable ion is a cation Diagram. 1 Fractionation with anion exchange chromatography An ionizable groups charged depends on the pH of the solvent and pKa of the ionizable group. IEX can be divided into two different sub types.
 Source: pt.pinterest.com
Source: pt.pinterest.com
Two distinct mechanisms as follows. Some of the charged regions which will influence ion exchange Interaction between opposite charges Charged groups on the proteins interact with charged groups on the ion exchanger. By varying the mobile phase concentration of the ion pair reagent. Ion Exchange Chromatography IEC or IEX is driven by interactions between charged target molecules and charged immobilized ligands on the chromatography resin. Salt Gradients As in most other modes of chromatography SEC being the exception a protein sample is injected onto the column under conditions where it will be strongly retained.
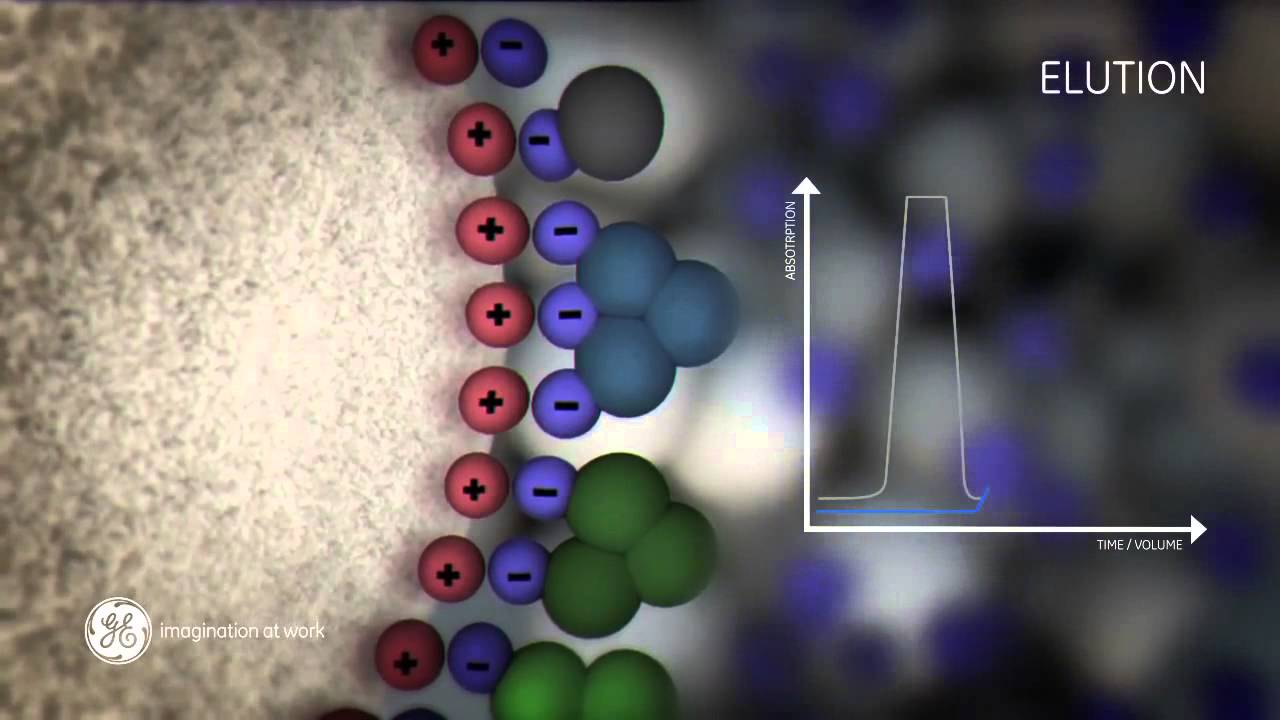 Source: in.pinterest.com
Source: in.pinterest.com
Water is made of water molecules formula H2O. The eluent is delivered to the system using a high-pressure pump. The sample is introduced then flows through the guard and into the analytical ion-exchange columns where the ion-exchange. Introduction In ion exchange chromatography retention is based on the attraction between the solute ions and charged sites bound to stationary phase Columns used for ion exchange are characterized by the presence of charged groups covalently attached to the stationary phase Anion exchangers contain bound positive groups where as cation. All natural waters contain some foreign substances usually in small amounts.
 Source: in.pinterest.com
Source: in.pinterest.com
In anion exchange chromatography a molecule with a pI of 68 may be run in a mobile phase buffer at pH 80 when the pKa of the solid support is 103. The purpose of adding an ion pair reagent to the mobile phase is usually to change the retention time of ionic analytes. By varying the mobile phase concentration of the ion pair reagent. Ion exchange chromatography is the reversible adsorption of charged molecules to immobilized ion groups on a matrix of an opposite charge. Ion Exchange chromatography Principle.
 Source: pinterest.com
Source: pinterest.com
Ion exchange has been the predominant form of ion chromatography to date 2. Ion exchange has been the predominant form of ion chromatography to date 2. Ion Exchange chromatography Principle. A typical ion chromatography consists of several components as shown in Figure 3. Rohm and Haas Ion Exchange Ion exchange introduction 1 FD Sep 2008 ION EXCHANGE FOR DUMMIES An introduction Water Water is a liquid.
 Source: pinterest.com
Source: pinterest.com
Ion Exchange Chromatography IEC or IEX is driven by interactions between charged target molecules and charged immobilized ligands on the chromatography resin. Rohm and Haas Ion Exchange Ion exchange introduction 1 FD Sep 2008 ION EXCHANGE FOR DUMMIES An introduction Water Water is a liquid. The eluent is delivered to the system using a high-pressure pump. Ion- exchange chromatography is based on electrostatic interactions between charged protein groups and solid support material matrix. Ion exchange starts with the equilibration of the exchanger using pH and ionic strength.
 Source: pinterest.com
Source: pinterest.com
Ion Exchange chromatography Principle. Ion exchange starts with the equilibration of the exchanger using pH and ionic strength. It is widely used as a preparative technique for Atomic Absorption Spectroscopy AAS Multiple Collector Inductively Coupled Plasma Mass. Rohm and Haas Ion Exchange Ion exchange introduction 1 FD Sep 2008 ION EXCHANGE FOR DUMMIES An introduction Water Water is a liquid. Ion chromatography includes all chromatographic methods that separate ionic substances and substances that dissociate easily.
 Source: pinterest.com
Source: pinterest.com
Ion exchange due to competitive ionic binding attraction and ion exclusion due to repulsion between similarly charged analyte ions and the ions fixed on the chromatographic support play a role in the separation in ion chromatography. Ion- exchange chromatography is based on electrostatic interactions between charged protein groups and solid support material matrix. In anion exchange chromatography a molecule with a pI of 68 may be run in a mobile phase buffer at pH 80 when the pKa of the solid support is 103. Schematic diagram of an anion exchange media particle. Rohm and Haas Ion Exchange Ion exchange introduction 1 FD Sep 2008 ION EXCHANGE FOR DUMMIES An introduction Water Water is a liquid.
This site is an open community for users to submit their favorite wallpapers on the internet, all images or pictures in this website are for personal wallpaper use only, it is stricly prohibited to use this wallpaper for commercial purposes, if you are the author and find this image is shared without your permission, please kindly raise a DMCA report to Us.
If you find this site helpful, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also bookmark this blog page with the title diagram of ion exchange chromatography by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.