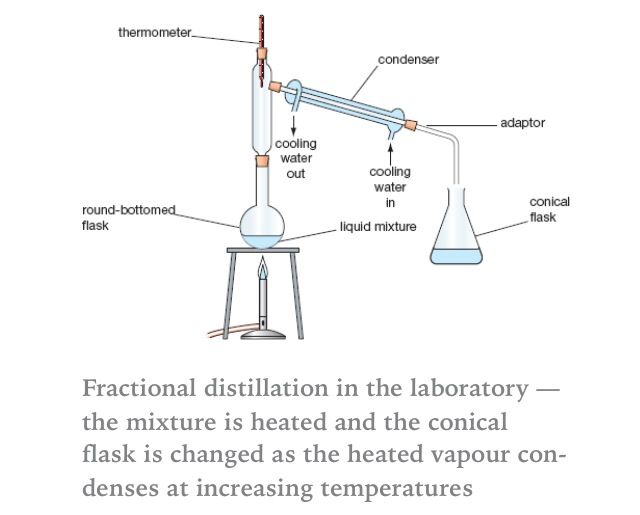
Diagram of simple distillation
Home » Background » Diagram of simple distillationYour Diagram of simple distillation images are ready in this website. Diagram of simple distillation are a topic that is being searched for and liked by netizens now. You can Get the Diagram of simple distillation files here. Get all royalty-free vectors.
If you’re looking for diagram of simple distillation pictures information linked to the diagram of simple distillation interest, you have come to the ideal blog. Our site frequently gives you hints for viewing the maximum quality video and picture content, please kindly hunt and find more informative video content and graphics that match your interests.
Diagram Of Simple Distillation. The simple distillation apparatus shown on page T 11 would only separate liquids of widely different boiling points. Click Share to make it public. How To Determine Boiling Point From Simple Distillation Graph. Show more Show less.
 New Bitesize Infographics Illustrating The Knowledge Learning Product Distillation Fractional Distillation Paper Chromatography From pinterest.com
New Bitesize Infographics Illustrating The Knowledge Learning Product Distillation Fractional Distillation Paper Chromatography From pinterest.com
This method works because water has a. To discuss distillation we need a temperature composition diagram pressure is held constant. Vapor Pressure is the pressure exerted by the vapor phase in equilibrium with the liquid phase. Solved Using The Phase Diagram Below Of Simple Distillati. Some examples of the use of simple distillation applied to various components are explained below. How To Determine Boiling Point From Simple Distillation Graph.
This method is used to separate the mixture of two miscible liquids where difference between their boiling points is at least 25 ºC.
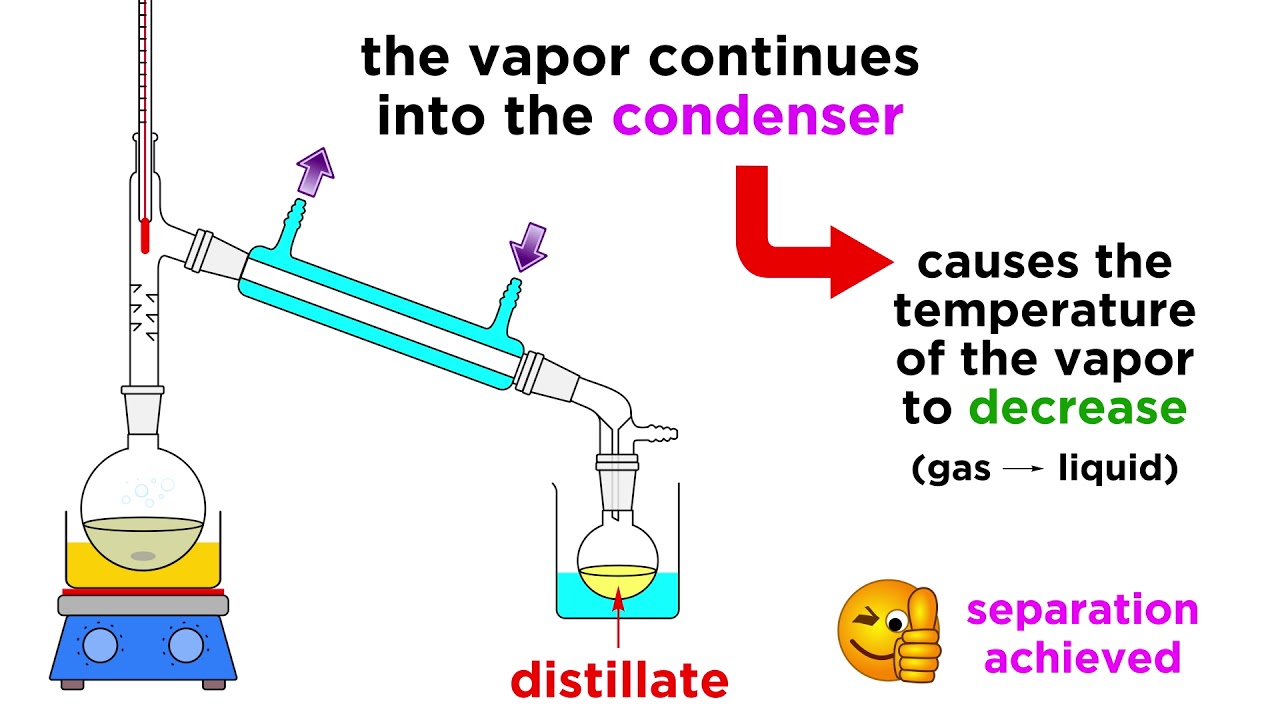
Familiar examples include the distillation of crude fermentation broths into alcoholic spirits such as gin and vodka and the fractionation of crude oil into useful products such as gasoline and heating oil. Batch Distillation Process is also called as Differential Distillation or Rayleigh Distillation. When the vapor enters the condenser whose walls are cooled by the jacket of cold water it cools and condenses back to a liquid called the distillate or condensate. DISTILLATION APPARATUS Distillation is a common operation in many laboratories for the purpose of separating andor purifying components of a liquid mixture. The vapor-liquid diagram is a plot relating the temperature of the vapor and liquid to the mole fraction of each component. The process of distillation begins with heating a liquid to boiling point.
 Source: pinterest.com
Source: pinterest.com
To discuss distillation we need a temperature composition diagram pressure is held constant. The apparatus used consists of three major parts. Vapor Pressure is the pressure exerted by the vapor phase in equilibrium with the liquid phase. Hence separation is feasible. Batch Distillation Process is also called as Differential Distillation or Rayleigh Distillation.
 Source: pinterest.com
Source: pinterest.com
At a given temperature the composition of the liquid and vapor phases can be determined. Simple distillation works because the dissolved solute. DISTILLATION APPARATUS Distillation is a common operation in many laboratories for the purpose of separating andor purifying components of a liquid mixture. How is a vapor-liquid equilibrium diagram used to determine the composition of the liquid and vapor in a distillation. Explain the method of simple distillation.
 Source: pinterest.com
Source: pinterest.com
The Figure 1 setup consists of a container where a fixed quantity of the liquid mixture is fed initially. First we have the distillation of water and alcohol. This leaderboard is currently private. Click Share to make it public. Simple Distillation Diagram Showing The Simple Distillation Of A.
 Source: pinterest.com
Source: pinterest.com
Batch Distillation Process is also called as Differential Distillation or Rayleigh Distillation. Solved Using The Phase Diagram Below Of Simple Distillati. The liquid in the distillation flask is heated until it reaches its boiling point. The cooled vapor then condenses forming a distillate. Click Share to make it public.
 Source: pinterest.com
Source: pinterest.com
Click Share to make it public. Simple distillation works because the dissolved solute. Distillation is a unit operation employed to purify or to separate components in a mixture on the basis of their volatility. Vapor Pressure is the pressure exerted by the vapor phase in equilibrium with the liquid phase. This method is used to separate the mixture of two miscible liquids where difference between their boiling points is at least 25 ºC.
 Source: pinterest.com
Source: pinterest.com
As the name indicates this type of distillation is a batch process and not a continuous process. There are two types of phase diagram. This is represented by the solid line in the diagram of the distillation curves. More volatile component vaporizes faster than less volatile component. 5 shows such a diagram for 2 solvents A and B.
 Source: pinterest.com
Source: pinterest.com
The process of distillation begins with heating a liquid to boiling point. For example water can be separated from salt solution by simple distillation. Phase Diagram For binary mixture phase diagram only two-component mixture eg. 5 shows such a diagram for 2 solvents A and B. Acetone and water are miscible liquids also the difference between their boiling point is more than 25 ºC so they can be separated by the method of simple distillation.
 Source: pinterest.com
Source: pinterest.com
There are two types of phase diagram. Familiar examples include the distillation of crude fermentation broths into alcoholic spirits such as gin and vodka and the fractionation of crude oil into useful products such as gasoline and heating oil. A more volatile and B less volatile are considered. Process Flow Diagram Symbols. Distillation is a unit operation employed to purify or to separate components in a mixture on the basis of their volatility.
 Source: pinterest.com
Source: pinterest.com
To understand the nature of simple distillation fractional distillation and azeotropes we need to look at vaporliquid diagrams for pairs of solvents. Simple distillation is used to separate a solvent from a solution. Distillation Introduction And Phase Diagram. The Boiling Point Diagram Separation Technologies. Simple distillation works because the dissolved solute.
 Source: pinterest.com
Source: pinterest.com
This is represented by the solid line in the diagram of the distillation curves. The graph below Fig. First you have a solution of alcohol in water that has a concentration of 50. If a separation were attempted of two liquids with similar boiling points or if a. This is represented by the solid line in the diagram of the distillation curves.
 Source: pinterest.com
Source: pinterest.com
Click Share to make it public. Solved Using The Phase Diagram Below Of Simple Distillati. Revision notes on simple distillation diagram fractional distillation apparatus explained help when revising for AQA GCSE 9-1 chemistry Edexcel GCSE 9-1 chemistry OCR GCSE 9-1 gateway science OCR GCSE 9-1 21st century science GCSE 9-1. Spray Drying Distilled Water Purified Water Diagram Png. Some examples of the use of simple distillation applied to various components are explained below.
 Source: pinterest.com
Source: pinterest.com
This method works because water has a. T Xy And Xy Diagrams For The Binary Mixture Acetone Water A Pinch. The bottom of the graph shows the liquid state and the top of the graph shows the vapor state. In this case the boiling point of the alcohol is known to be 784 C. As the name indicates this type of distillation is a batch process and not a continuous process.
 Source: pinterest.com
Source: pinterest.com
The liquid in the distillation flask is heated until it reaches its boiling point. How To Determine Boiling Point From Simple Distillation Graph. Simple distillation is used to separate a solvent from a solution. This method works because water has a. Batch Distillation Process is also called as Differential Distillation or Rayleigh Distillation.
 Source: pinterest.com
Source: pinterest.com
Distillation is a unit operation employed to purify or to separate components in a mixture on the basis of their volatility. The process of distillation begins with heating a liquid to boiling point. Distillation Introduction And Phase Diagram. The hot vapors above the boiling liquid rise through the distillation head and enter the condenser. To understand the nature of simple distillation fractional distillation and azeotropes we need to look at vaporliquid diagrams for pairs of solvents.
 Source: pinterest.com
Source: pinterest.com
Place pure liquid can be obtained. This is represented by the solid line in the diagram of the distillation curves. It is useful for producing water from salt solution. The simple distillation apparatus shown on page T 11 would only separate liquids of widely different boiling points. The cooled vapor then condenses forming a distillate.
 Source: in.pinterest.com
Source: in.pinterest.com
The process of distillation begins with heating a liquid to boiling point. Distillation Distillation is a commonly used method for purifying liquids and separating mixtures of liquids into their individual components. The vapor is then cooled usually by passing it through pipes or tubes at a lower temperature. There are two types of phase diagram. This process flow diagram PFD of a typical crude oil distillation unit as used in petroleum crude oil refineries was redrawn from Wikipedia file.
 Source: pinterest.com
Source: pinterest.com
5 shows such a diagram for 2 solvents A and B. How is a vapor-liquid equilibrium diagram used to determine the composition of the liquid and vapor in a distillation. To discuss distillation we need a temperature composition diagram pressure is held constant. When the vapor enters the condenser whose walls are cooled by the jacket of cold water it cools and condenses back to a liquid called the distillate or condensate. Batch Distillation Process is also called as Differential Distillation or Rayleigh Distillation.
 Source: pinterest.com
Source: pinterest.com
Place pure liquid can be obtained. Distillation Introduction And Phase Diagram. Simple Distillation Fractional Distillation Method Of Separating A. Spray Drying Distilled Water Purified Water Diagram Png. Crude Oil Distillation Unitpng.
This site is an open community for users to do submittion their favorite wallpapers on the internet, all images or pictures in this website are for personal wallpaper use only, it is stricly prohibited to use this wallpaper for commercial purposes, if you are the author and find this image is shared without your permission, please kindly raise a DMCA report to Us.
If you find this site adventageous, please support us by sharing this posts to your favorite social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title diagram of simple distillation by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.