Draw diagrams to illustrate condensation and hydrolysis reactions
Home » Background » Draw diagrams to illustrate condensation and hydrolysis reactionsYour Draw diagrams to illustrate condensation and hydrolysis reactions images are ready. Draw diagrams to illustrate condensation and hydrolysis reactions are a topic that is being searched for and liked by netizens today. You can Get the Draw diagrams to illustrate condensation and hydrolysis reactions files here. Find and Download all free images.
If you’re searching for draw diagrams to illustrate condensation and hydrolysis reactions images information related to the draw diagrams to illustrate condensation and hydrolysis reactions keyword, you have visit the ideal site. Our website always provides you with suggestions for seeking the maximum quality video and image content, please kindly hunt and find more enlightening video articles and graphics that match your interests.
Draw Diagrams To Illustrate Condensation And Hydrolysis Reactions. The much weaker bonding between base pairs caused by hydrogen bonds does not involve the sort of condensation reaction already mentioned. A large molecule is split into smaller sections by breaking a bond adding -H to one section and -OH to the other. Hydrolysis reaction is the reverse of condensation reaction which bonds are broken by the addition of water molecules. Distinguish between monomers and polymers.
 Hydrolysis Definition Reaction Equation Example Study Com From study.com
Hydrolysis Definition Reaction Equation Example Study Com From study.com
Monomer polymer condensation reaction aka-dehydration reaction and. Draw diagrams to illustrate condensation and hydrolysis reactions. Distinguish between monomers and polymers. The diagram below by identifying a. Monomers are connected by condensation reactions. Complete the following table below by filling in the atoms found in each biomolecule aka macromolecule.
The Molecules of Life List the four major classes of macromolecules.
C Draw diagrams to illustrate condensation and hydrolysis reactions. Draw diagrams to illustrate condensation and hydrolysis reactions. A condensation reaction occurs when two molecules join to form a larger molecule and release a smaller molecule s in the process. C Distinguish between the glycosidic linkages found in starch and cellulose. IV Carbohydrates Serve as Fuel and Building Material A Distinguish among monosaccharides disaccharides and polysaccharides. Hydrolysis is the process of breaking a molecule into two parts using water.
 Source: pinterest.com
Source: pinterest.com
Ester Hydrolysis Reaction If we take the same example as in dehydration synthesis reaction hydrolysis of an ester forms the reactants of esterification reaction. IV Carbohydrates Serve as Fuel and Building Material A Distinguish among monosaccharides disaccharides and polysaccharides. Condensation reaction - a reaction in which two molecules become covalently bonded to each other though the loss of a small molecule usually water. The diagram below by identifying a. The Molecules of Life List the four major classes of macromolecules.
 Source: pinterest.com
Source: pinterest.com
Condensation polymerizationa polymerization mechanism in which monomers react to form dimers first then trimers longer oligomers and eventually long chain polymers. The smaller molecule lost in the reaction is often water but it can also be methanol hydrogen chloride acetic acid or several other molecules. Monomers are connected by a reaction in which two molecules are covalently. Distinguish between monosaccharides disaccharides and polysaccharides. Describe the formation of a glycosidic linkage and distinguish between the glycosidic linkages found in starch and cellulose.
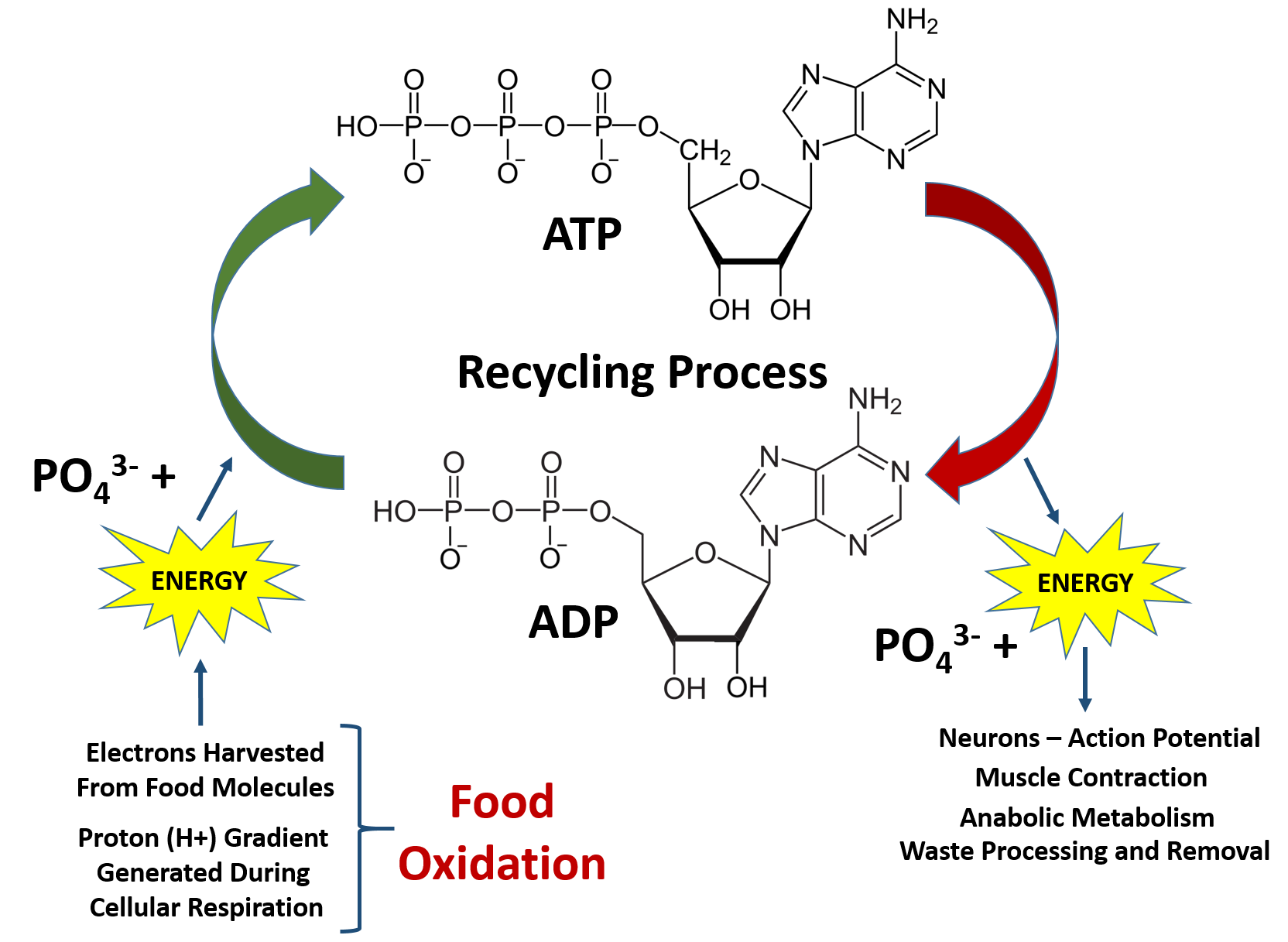 Source: wou.edu
Source: wou.edu
The Molecules of Life List the four major classes of macromolecules. Hydrolysis reaction is the reverse of condensation reaction which bonds are broken by the addition of water molecules. Monomers are connected by condensation reactions. Carbohydrates Serve as Fuel and Building Material Distinguish between monosaccharides disaccharides and polysaccharides. Complete the following table below by filling in the atoms found in each biomolecule aka macromolecule.
 Source: pinterest.com
Source: pinterest.com
Hydrolysis reaction is the reverse of condensation reaction which bonds are broken by the addition of water molecules. The smaller molecule lost in the reaction is often water but it can also be methanol hydrogen chloride acetic acid or several other molecules. A large molecule is split into smaller sections by breaking a bond adding -H to one section and -OH to the other. C Distinguish between the glycosidic linkages found in starch and cellulose. Distinguish between monomers and polymers.
 Source: pinterest.com
Source: pinterest.com
Condensation reactions are reactions between two molecules that create one large and one small usually H 2 O molecule. Hydrolysis is reverse of condensation. Ester Hydrolysis Reaction If we take the same example as in dehydration synthesis reaction hydrolysis of an ester forms the reactants of esterification reaction. Learn more about the definition of hydrolysis and a hydrolysis reaction including how acids and. The diagram below by identifying a.
 Source: quizlet.com
Source: quizlet.com
Also known as condensation reaction dehydration synthesis is the process of combining small molecules called monomers in order to produce larger units called polymers following the removal of water H 2. Hydrolysis is the process of breaking a molecule into two parts using water. Choose from nitrogen N carbon C phosphorus P oxygen O and hydrogen H Macromolecule Atoms Found in Biomolecule. Polymers are disassembled by hydrolysis. Distinguish between monosaccharides disaccharides and polysaccharides.
 Source: study.com
Source: study.com
Hydrolysis reactions use water to break up another molecule usually by splitting up the water into H and OH ions. Describe the formation of a glycosidic linkage and distinguish between the glycosidic linkages found in starch and cellulose. Distinguish between monosaccharides disaccharides and polysaccharides. In a hydrolysis reaction involving an ester link such as that found between two amino acids in a protein the molecule is split. Polymers are disassembled by hydrolysis.
 Source: pinterest.com
Source: pinterest.com
Hydrolysis - basic principles Hydrolysis is the opposite to condensation. B Describe the formation of a glycosidic linkage. Distinguish between monosaccharides disaccharides and polysaccharides. Distinguish between the glycosidic linkages found in starch and cellulose. Draw diagrams to illustrate condensation and hydrolysis reactions.
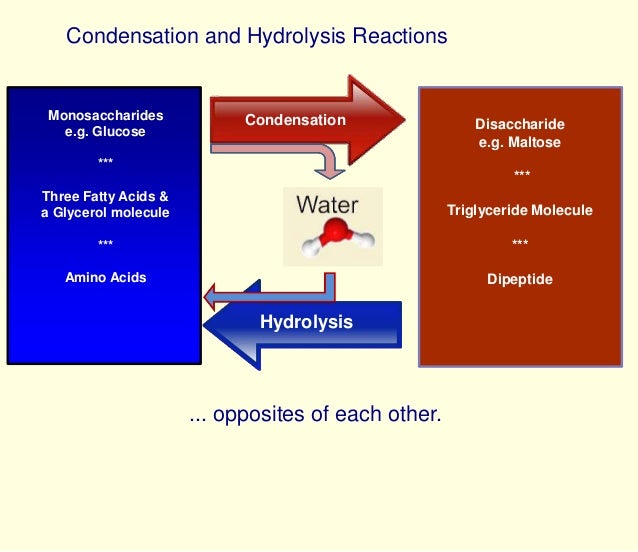 Source: slideshare.net
Source: slideshare.net
Draw diagrams to illustrate condensation and hydrolysis reactions. Hydrolysis reaction is the reverse of condensation reaction which bonds are broken by the addition of water molecules. In a hydrolysis reaction involving an ester link such as that found between two amino acids in a protein the molecule is split. For example in the hydrolysis of ethyl acetate an ester reacts with water to produce acetic acid and ethanol. C Distinguish between the glycosidic linkages found in starch and cellulose.
 Source: pinterest.com
Source: pinterest.com
Hydrolysis is reverse of condensation. Monomer polymer condensation reaction aka-dehydration reaction and. C Draw diagrams to illustrate condensation and hydrolysis reactions. A large molecule is split into smaller sections by breaking a bond adding -H to one section and -OH to the other. The Molecules of Life List the four major classes of macromolecules.
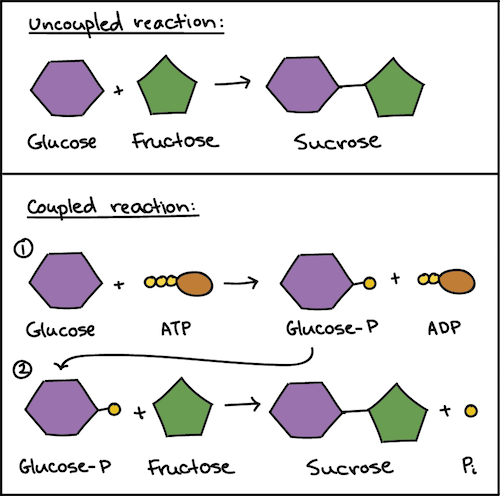 Source: khanacademy.org
Source: khanacademy.org
Condensation reactions occur when two compounds react and produce water as a by-product. Condensation polymerizationa polymerization mechanism in which monomers react to form dimers first then trimers longer oligomers and eventually long chain polymers. Condensation reactions occur when two compounds react and produce water as a by-product. In a hydrolysis reaction involving an ester link such as that found between two amino acids in a protein the molecule is split. The products are simpler substances.
 Source: pinterest.com
Source: pinterest.com
Condensation reaction - a reaction in which two molecules become covalently bonded to each other though the loss of a small molecule usually water. Carbohydrates Serve as Fuel and Building Material. The formal reverse of hydrolysis. Draw diagrams to illustrate condensation and hydrolysis reactions. Condensation reaction is dehydration reaction.
 Source: pinterest.com
Source: pinterest.com
Carbohydrates Serve as Fuel and Building Material Distinguish between monosaccharides disaccharides and polysaccharides. C Distinguish between the glycosidic linkages found in starch and cellulose. B Describe the formation of a glycosidic linkage. Draw diagrams to illustrate condensation and hydrolysis reactions. For this module we will be learning about two condensation reactions.
 Source: pinterest.com
Source: pinterest.com
Distinguish between monomers and polymers. Monomers are connected by condensation reactions. Carbohydrates Serve as Fuel and Building Material Distinguish between monosaccharides disaccharides and polysaccharides. Distinguish between the glycosidic linkages found in starch and cellulose. Condensation reactions are reactions between two molecules that create one large and one small usually H 2 O molecule.
 Source: pinterest.com
Source: pinterest.com
IV Carbohydrates Serve as Fuel and Building Material A Distinguish among monosaccharides disaccharides and polysaccharides. The diagram below by identifying a. Draw diagrams to illustrate condensation and hydrolysis reactions. The smaller molecule lost in the reaction is often water but it can also be methanol hydrogen chloride acetic acid or several other molecules. Draw diagrams to illustrate condensation and hydrolysis reactions.
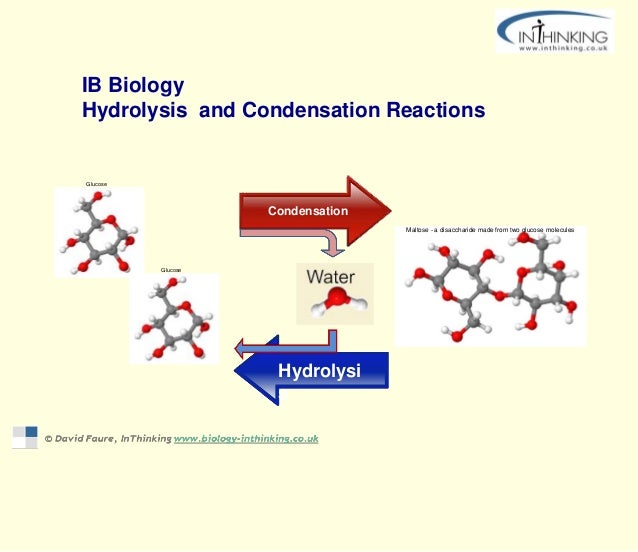 Source: slideshare.net
Source: slideshare.net
Hydrolysis - basic principles Hydrolysis is the opposite to condensation. Ester Hydrolysis Reaction If we take the same example as in dehydration synthesis reaction hydrolysis of an ester forms the reactants of esterification reaction. Distinguish between monosaccharides disaccharides and polysaccharides. C Draw diagrams to illustrate condensation and hydrolysis reactions. Distinguish between the glycosidic linkages found in starch and cellulose.
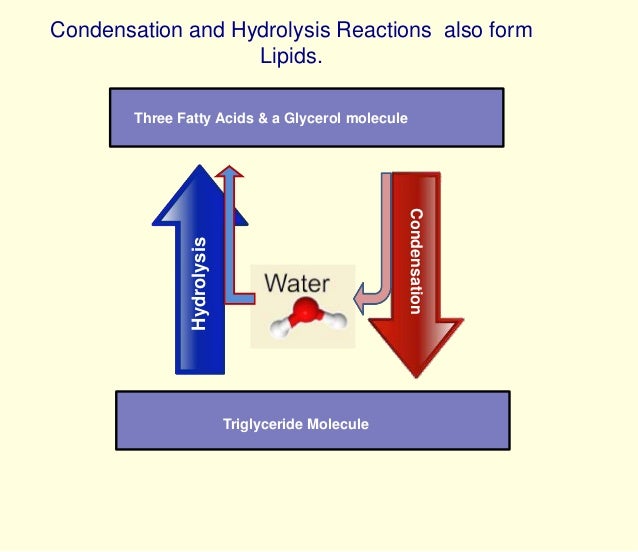 Source: slideshare.net
Source: slideshare.net
Condensation reactions occur when two compounds react and produce water as a by-product. Carbohydrates Serve as Fuel and Building Material Distinguish between monosaccharides disaccharides and polysaccharides. C Draw diagrams to illustrate condensation and hydrolysis reactions. Distinguish between the glycosidic linkages found in starch and cellulose. Draw diagrams to illustrate condensation and hydrolysis reactions.
 Source: pinterest.com
Source: pinterest.com
Condensation reactionany reaction in which two molecules react with the resulting loss of a water molecule or other small molecule. Distinguish between monosaccharides disaccharides and polysaccharides. The smaller molecule lost in the reaction is often water but it can also be methanol hydrogen chloride acetic acid or several other molecules. For example in the hydrolysis of ethyl acetate an ester reacts with water to produce acetic acid and ethanol. Monomers are connected by a reaction in which two molecules are covalently.
This site is an open community for users to share their favorite wallpapers on the internet, all images or pictures in this website are for personal wallpaper use only, it is stricly prohibited to use this wallpaper for commercial purposes, if you are the author and find this image is shared without your permission, please kindly raise a DMCA report to Us.
If you find this site convienient, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also bookmark this blog page with the title draw diagrams to illustrate condensation and hydrolysis reactions by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.