Electron dot diagram calculator
Home » Background » Electron dot diagram calculatorYour Electron dot diagram calculator images are available in this site. Electron dot diagram calculator are a topic that is being searched for and liked by netizens now. You can Download the Electron dot diagram calculator files here. Get all royalty-free photos and vectors.
If you’re searching for electron dot diagram calculator pictures information linked to the electron dot diagram calculator keyword, you have pay a visit to the right site. Our site frequently provides you with suggestions for downloading the highest quality video and picture content, please kindly search and find more informative video articles and images that match your interests.
Electron Dot Diagram Calculator. They consist of the symbol for the element in the center surrounded by dots indicating the number of valence electrons. Find the required count of electrons needed to make the atoms complete. This is the common convention we will adopt although there will be exceptions later. The drawing only works f0r stable molecules that actually exist.
 Co2 Lewis Structure Easy Hard Science From learnwithdrscott.com
Co2 Lewis Structure Easy Hard Science From learnwithdrscott.com
To generate the Lewis dot structure you have to follow the given steps. Clickingtapping the hot spot opens the Concept Builder in full-screen mode. To embed a widget in your blogs sidebar install the WolframAlpha Widget Sidebar Plugin and copy and paste the Widget ID below into the id field. Lewis dot structure for CO HNO3C2H4 H2S Chemistry. In the first step we will calculate how many valence electrons are present for CHCl3 so that we can arrange them in the lewis diagram. To include the widget in a wiki page paste the code.
This is the common convention we will adopt although there will be exceptions later.
So if you type that structure into google you should receive the lewis structure. Lewis dot structure for CO HNO3C2H4 H2S Chemistry. For this look at the periodic group of each atom. Elements in the same column of the periodic table have similar Lewis electron dot diagrams because they have the same valence shell electron configuration. Ozone consists of three oxygen atoms. They are similar to electron dot diagrams in that the valence electrons in lone pairs.

Electron Configuration Calculator Added Nov 11 2014 by Brennenlb in Chemistry Find the Electron configuration of any Element on the Periodic Table of Elements with this simple yet very useful widget. For this look at the periodic group of each atom. Use the Escape key on a keyboard or comparable method to exit from full-screen mode. Here we will be dealing with ozone the molecular formula is O3. Lewis dot structure of C 2 H 6.
 Source: learnwithdrscott.com
Source: learnwithdrscott.com
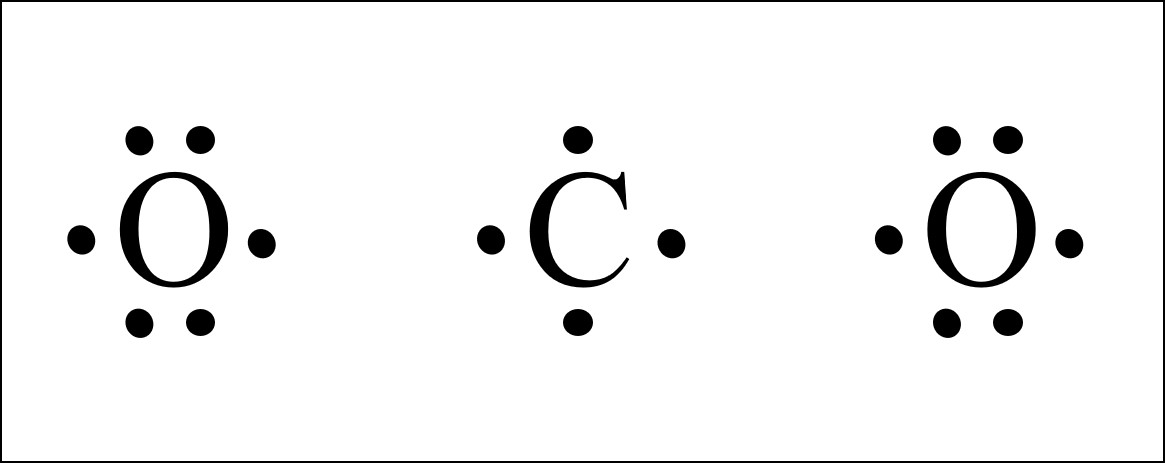
The resulting Lewis electron dot structure displays a triple bond connecting a carbon and an oxygen atom each holding a lone pair of electrons. Hence total shared pairs of electrons in the dot diagram. Calculate the total valence electrons in the molecule. Count total valence electron in CHCl3. In this step add the total count of valence electrons from all the atoms in a bit.
 Source: shimizu-uofsc.net
Source: shimizu-uofsc.net
The number of dots equals the number of valence electrons in the atom. Clickingtapping the hot spot opens the Concept Builder in full-screen mode. The next atom lithium has an electron configuration of 1s 2 2s 1 so it has only one electron in its valence shellIts electron dot diagram resembles that of hydrogen except. One potential electron dot formula for the carbonate ion is. For example carbon has four valence electrons and the symbol C so it.
 Source: courses.lumenlearning.com
Source: courses.lumenlearning.com
Lewis electron dot diagrams for ions have less for. For example carbon has four valence electrons and the symbol C so it. Elements in the same column of the periodic table have similar Lewis electron dot diagrams because they have the same valence shell electron configuration. It thus has 6 valence electrons. This is a series of lectures in videos covering Chemistry topics taught in schools.

Find the total count of valence electrons to molecules. Dot diagrams are very different to orbital diagrams but theyre still very easy to understand. This is the common convention we will adopt although there will be exceptions later. In the first step we will calculate how many valence electrons are present for CHCl3 so that we can arrange them in the lewis diagram. Valence electron of carbon 4 Bonding electrons 8.
 Source: chem.libretexts.org
Source: chem.libretexts.org
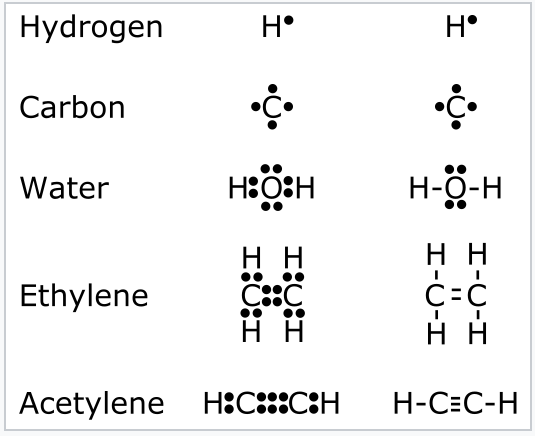
A Lewis electron dot diagram or electron dot diagram or a Lewis diagram or a Lewis structure is a representation of the valence electrons of an atom that uses dots around the symbol of the element. To generate the Lewis dot structure you have to follow the given steps. They consist of the symbol for the element in the center surrounded by dots indicating the number of valence electrons. By putting the two electrons together on the same side we emphasize the fact that these two electrons are both in the 1s subshell. There is a second hot-spot in the lower-right corner.
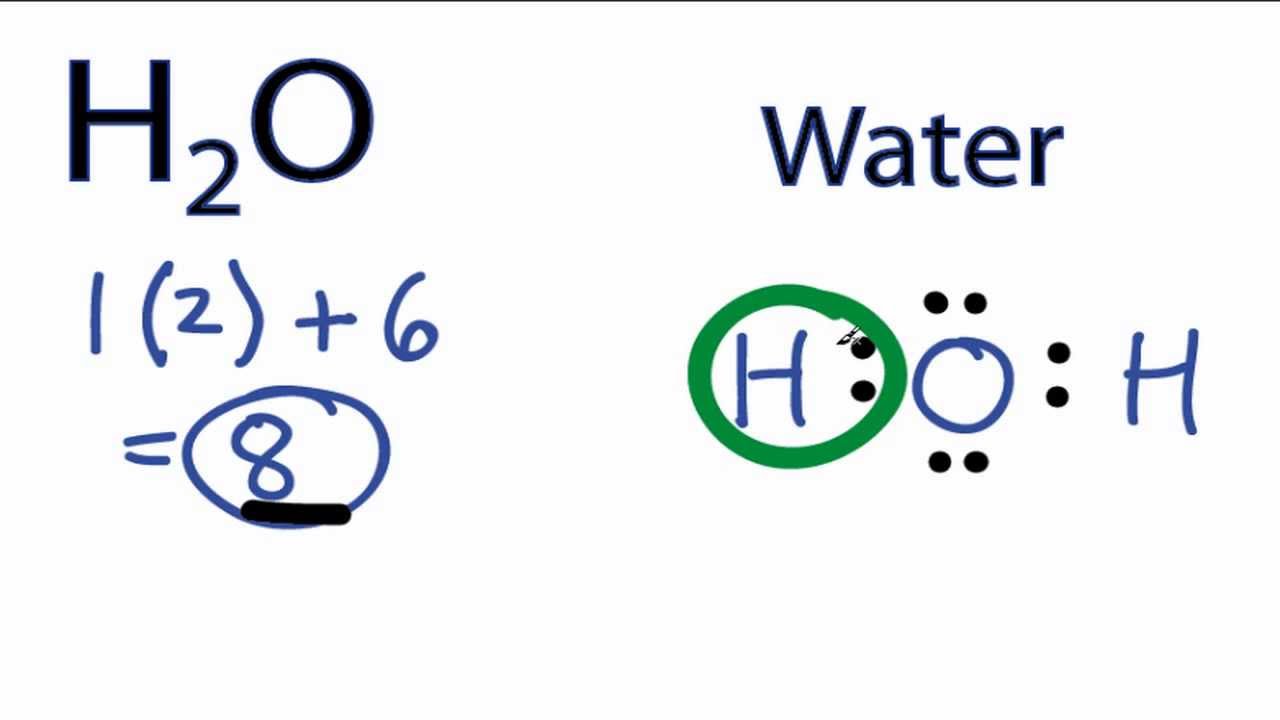 Source: youtube.com
Source: youtube.com
The lewis structure is used to represent the covalent bonding of a molecule or ion. There is a second hot-spot in the lower-right corner. Given below is the image of the molecular orbital diagram of H3O and also that of H2O for reference. Clickingtapping the hot spot opens the Concept Builder in full-screen mode. Ozone consists of three oxygen atoms.
 Source: youtube.com
Source: youtube.com
Valence electron of carbon 4 Bonding electrons 8. In terms of electron dot formulas define the electron structure of the carbonate ion CO 3 2-. So if you type that structure into google you should receive the lewis structure. The next atom lithium has an electron configuration of 1s 2 2s 1 so it has only one electron in its valence shellIts electron dot diagram resembles that of hydrogen except. It does not matter what order the positions are used For example the Lewis electron dot diagram for calcium is simply.
 Source: study.com
Source: study.com
It does not matter what order the positions are used For example the Lewis electron dot diagram for calcium is simply. For atoms with partially filled d or f subshells these electrons are typically omitted from Lewis electron dot diagrams. By putting the two electrons together on the same side we emphasize the fact that these two electrons are both in the 1s subshell. For this look at the periodic group of each atom. Calculate the total valence electrons in the molecule.
 Source: chemistrysteps.com
Source: chemistrysteps.com
After then define the number of bonds in the given molecule. To embed a widget in your blogs sidebar install the WolframAlpha Widget Sidebar Plugin and copy and paste the Widget ID below into the id field. In terms of electron dot formulas define the electron structure of the carbonate ion CO 3 2-. Lewis dot structure of C 2 H 6. B Identify the chemical formula of the ionic compound formed from the metal and the nonmetal based on your answer to part a.
 Source: youtube.com
Source: youtube.com
A beryllium atom with two valence electrons would have the electron dot diagram below. The number of dots equals the number of valence electrons in the atom. One potential electron dot formula for the carbonate ion is. This is the common convention we will adopt although there will be exceptions later. A Mg b Mg with ions that have an electron configuration identical to that of argon.
 Source: shimizu-uofsc.net
Source: shimizu-uofsc.net
The resulting Lewis electron dot structure displays a triple bond connecting a carbon and an oxygen atom each holding a lone pair of electrons. The resulting Lewis electron dot structure displays a triple bond connecting a carbon and an oxygen atom each holding a lone pair of electrons. The following lesson looks at drawing electron dot diagrams. If playback doesnt begin shortly try restarting your device. A Lewis electron dot diagram or electron dot diagram or a Lewis diagram or a Lewis structure is a representation of the valence electrons of an atom that uses dots around the symbol of the element.
 Source: dummies.com
Source: dummies.com
The number of dots equals the number of valence electrons in the atom. Electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the elements symbol. So its a nice tool to explore how atoms bond into more complex substances. For this look at the periodic group of each atom. Here we will be dealing with ozone the molecular formula is O3.
 Source: chem.libretexts.org
Source: chem.libretexts.org
The drawing only works f0r stable molecules that actually exist. For atoms with partially filled d or f subshells these electrons are typically omitted from Lewis electron dot diagrams. By putting the two electrons together on the same side we emphasize the fact that these two electrons are both in the 1s subshell. Lewis Structure of O3. For example carbon has four valence electrons and the symbol C so it.
 Source: study.com
Source: study.com
It does not matter what order the positions are used For example the Lewis electron dot diagram for calcium is simply. In the first step we will calculate how many valence electrons are present for CHCl3 so that we can arrange them in the lewis diagram. Lewis Structure of O3. Lewis Dot Structure Calculator - fasrplace. To embed a widget in your blogs sidebar install the WolframAlpha Widget Sidebar Plugin and copy and paste the Widget ID below into the id field.
 Source: studylib.net
Source: studylib.net
This is a series of lectures in videos covering Chemistry topics taught in schools. Total Watch the video of Dr. The next atom lithium has an electron configuration of 1s 2 2s 1 so it has only one electron in its valence shellIts electron dot diagram resembles that of hydrogen except. Count total valence electron in CHCl3. In this step add the total count of valence electrons from all the atoms in a bit.
 Source: pinterest.com
Source: pinterest.com
So if you type that structure into google you should receive the lewis structure. This is a series of lectures in videos covering Chemistry topics taught in schools. Follow some steps for drawing the lewis dot structure of CHCl3. One potential electron dot formula for the carbonate ion is. Dot diagrams are very different to orbital diagrams but theyre still very easy to understand.
 Source: slidesharetips.blogspot.com
Source: slidesharetips.blogspot.com
As many of the Lewis Dot Structures as you can before coming to lab. Find the required count of electrons needed to make the atoms complete. It does not matter what order the positions are used For example the Lewis electron dot diagram for calcium is simply. For atoms with partially filled d or f subshells these electrons are typically omitted from Lewis electron dot diagrams. Dot diagrams are very different to orbital diagrams but theyre still very easy to understand.
This site is an open community for users to do submittion their favorite wallpapers on the internet, all images or pictures in this website are for personal wallpaper use only, it is stricly prohibited to use this wallpaper for commercial purposes, if you are the author and find this image is shared without your permission, please kindly raise a DMCA report to Us.
If you find this site value, please support us by sharing this posts to your favorite social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title electron dot diagram calculator by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.