Electron transfer diagram
Home » Background » Electron transfer diagramYour Electron transfer diagram images are ready in this website. Electron transfer diagram are a topic that is being searched for and liked by netizens now. You can Get the Electron transfer diagram files here. Download all royalty-free vectors.
If you’re searching for electron transfer diagram images information related to the electron transfer diagram topic, you have pay a visit to the ideal site. Our site always gives you suggestions for seeking the highest quality video and image content, please kindly search and locate more enlightening video content and graphics that match your interests.
Electron Transfer Diagram. It is composed of cytochrome b c and a specific Fe-S center known as cytochrome reductase. Electron-transfer dissociation is a method of fragmenting multiply-charged gaseous macromolecules in a mass spectrometer between the stages of tandem mass spectrometry. The electrons begin their reactions in Complex I continuing onto Complex II traversed to Complex III and cytochrome c via coenzyme Q and then finally to Complex IV. The electron transport chain is made up of a series of spatially separated enzyme complexes that transfer electrons from electron donors to electron receptors via sets of redox reactions.
 6 1 Lewis Electron Dot Diagrams Introductory Chemistry From courses.lumenlearning.com
6 1 Lewis Electron Dot Diagrams Introductory Chemistry From courses.lumenlearning.com
Complex III catalyzes the transfer of two electrons from CoQH 2 to cytochrome c. Many of the ions that form have eight electrons in their valence shell. Either atoms gain enough electrons to have eight electrons in the valence shell and become the. The example below shows the transfer of a proton from the relatively acidic acetic acid molecule to the relatively basic hydroxide anion. These are cyclic and non- cyclic photophosphorylation Table 13-3. The eight valence electrons a full outer s and p sublevel give the noble gases their special stability.
In both cases the system is represented in two states that before electron transfer R the reactant state and that after electron transfer P the product state.
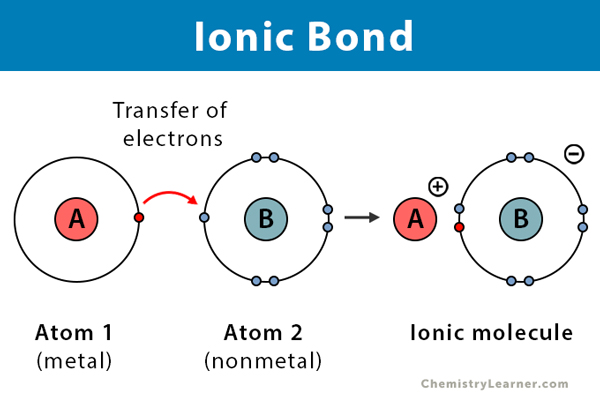
This step results in the translocation of four protons similar to complex I across. Demonstrate electron transfer between atoms to form ionic bonds. Chemists use simple diagrams to show an atoms valence electrons and how they transfer. Electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the elements symbol. In this process water splits into hydrogen ions oxygen and both ATP and NADPH 2 are produced. The general reaction coordinate diagram for the mechanism involved outer-sphere electron transfer reactions.
 Source: savemyexams.co.uk
Source: savemyexams.co.uk
In electron transfer the number of electrons lost must equal the number of electrons gained. This leads to the development of an electrochemical proton gradient across the membrane that activates the. The electron transport system refers to the Electron transport chain or ETS in abbreviated form that is present in the inner mitochondrial membraneETS involves electron transfer through a series of protein complexes from higher NADH to lower energy state O 2 by releasing protons into the cytosol. We show this process with one arrow labeled a in the diagram that starts at a lone pair of electrons on the basic. Similar to electron-capture dissociation ETD induces fragmentation of large multiply-charged cations by transferring electrons to them.
 Source: courses.lumenlearning.com
Source: courses.lumenlearning.com
ETD is used extensively with polymers and biological molecules such as proteins and peptides for sequence analysis. NAD H FMN NAD FMNH 2. Complex III Cytochrome bc1 complex. Some bacterial electron transport chains resemble the mitochondrial electron transport chain. FMN Flavin Mononucleotide At the beginning of the electron transfer chain the electrons from NADH are transferred to the flavin Mononucleotide FMN reducing it to FMNH 2.
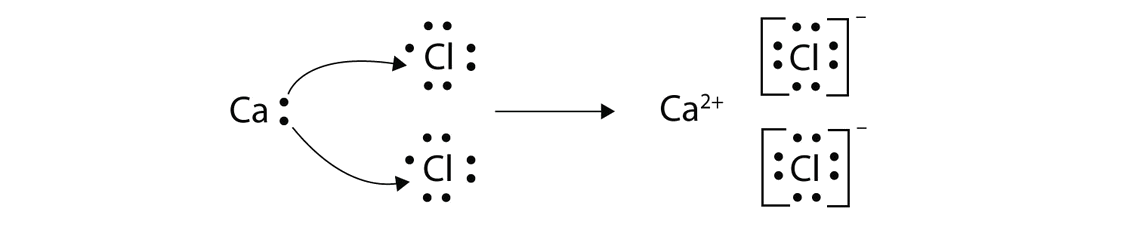
 Source: edurev.in
Source: edurev.in
In this process water splits into hydrogen ions oxygen and both ATP and NADPH 2 are produced. Electron-transfer dissociation is a method of fragmenting multiply-charged gaseous macromolecules in a mass spectrometer between the stages of tandem mass spectrometry. In electron transfer the number of electrons lost must equal the number of electrons gained. Similar to electron-capture dissociation ETD induces fragmentation of large multiply-charged cations by transferring electrons to them. There are about 15 different chemical groups that accept or transfer electrons through the electron chain.
 Source: courses.lumenlearning.com
Source: courses.lumenlearning.com
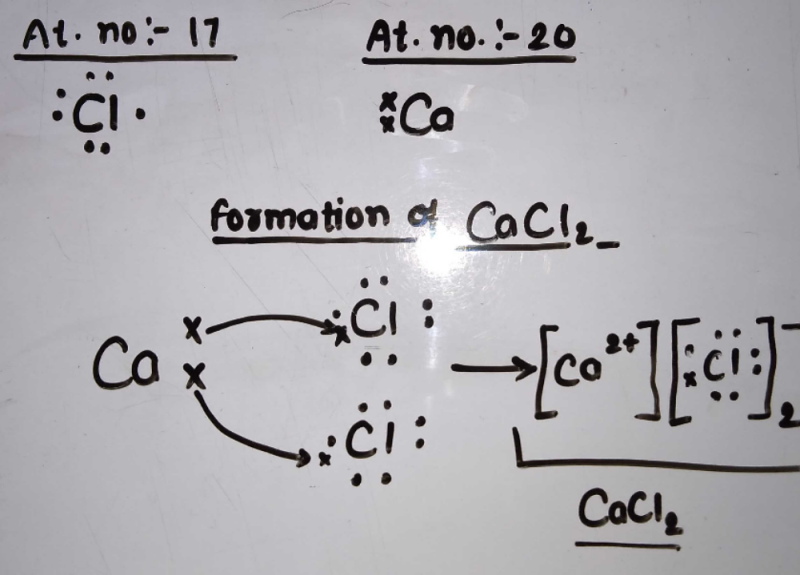
Demonstrate electron transfer between atoms to form ionic bonds. Paracoccus denitrificans is a gram-negative facultative anaerobic soil. The example below shows the transfer of a proton from the relatively acidic acetic acid molecule to the relatively basic hydroxide anion. Complex III catalyzes the transfer of two electrons from CoQH 2 to cytochrome c. These diagrams have two advantages over the electron shell diagrams introduced in Chapter 2 Elements Atoms and the Periodic Table.
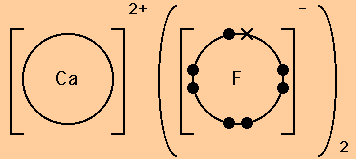 Source: legacy.chemgym.net
Source: legacy.chemgym.net
Secondly ET is rather simple and very suitable to be used as a model for studying solvent effects and to relate the kinetics of ET reactions to. Transfer of Electrons from CoQH 2 to Cytochrome c. The astute reader may have noticed something. Some bacterial electron transport chains resemble the mitochondrial electron transport chain. Many of the ions that form have eight electrons in their valence shell.
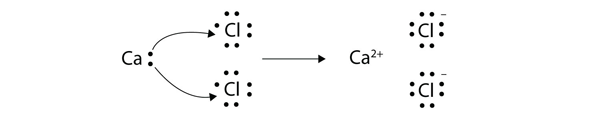
 Source: chem.libretexts.org
Source: chem.libretexts.org
Showing the transfer of electrons that takes place between metals and nonmetal atoms - thus forming oppositely charged ions. Chemists use simple diagrams to show an atoms valence electrons and how they transfer. It is composed of cytochrome b c and a specific Fe-S center known as cytochrome reductase. ETD is used extensively with polymers and biological molecules such as proteins and peptides for sequence analysis. The electrostatic attraction.
 Source: texasgateway.org
Source: texasgateway.org
The general reaction coordinate diagram for the mechanism involved outer-sphere electron transfer reactions. These diagrams have two advantages over the electron shell diagrams introduced in Chapter 2 Elements Atoms and the Periodic Table. Chemists use simple diagrams to show an atoms valence electrons and how they transfer. In the transition state of the complex the electrons have equal probability. Many of the ions that form have eight electrons in their valence shell.

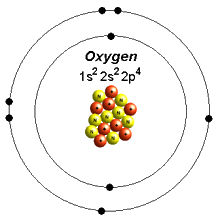
NAD H FMN NAD FMNH 2. It is composed of cytochrome b c and a specific Fe-S center known as cytochrome reductase. In Section 91 Lewis Electron Dot Diagrams we saw how ions are formed by losing electrons to make cations or by gaining electrons to form anions. Complex III catalyzes the transfer of two electrons from CoQH 2 to cytochrome c. The number of dots equals the number of valence electrons in the atom.
 Source: chemistrylearner.com
Source: chemistrylearner.com
The example below shows the transfer of a proton from the relatively acidic acetic acid molecule to the relatively basic hydroxide anion. FMN Flavin Mononucleotide At the beginning of the electron transfer chain the electrons from NADH are transferred to the flavin Mononucleotide FMN reducing it to FMNH 2. ETD is used extensively with polymers and biological molecules such as proteins and peptides for sequence analysis. The electrostatic attraction. The electron transport chain is made up of a series of spatially separated enzyme complexes that transfer electrons from electron donors to electron receptors via sets of redox reactions.
 Source: itc.gsw.edu
Source: itc.gsw.edu
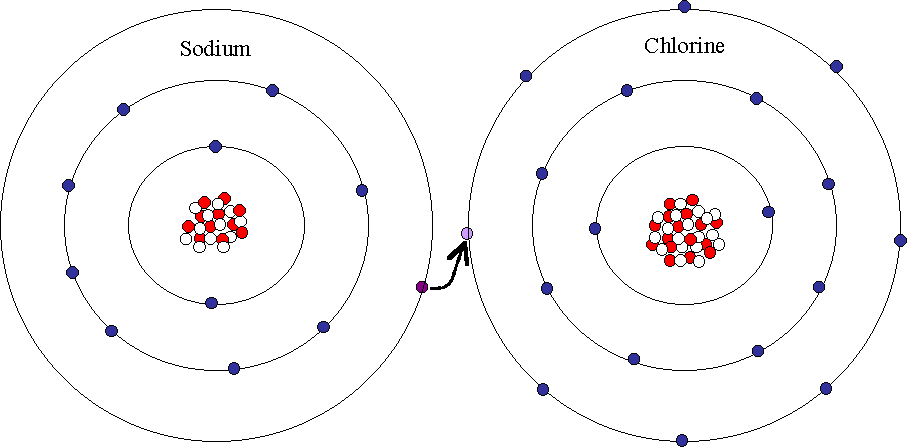
The course of the complete reaction is shown below. A Lewis electron dot diagram or electron dot diagram or a Lewis diagram or a Lewis structure is a representation of the valence electrons of an atom that uses dots around the symbol of the element. We saw this in the formation of NaCl. Similar to electron-capture dissociation ETD induces fragmentation of large multiply-charged cations by transferring electrons to them. In the section Lewis Electron Dot Diagrams we saw how ions are formed by losing electrons to make cations or by gaining electrons to form anions.
 Source: techiescientist.com
Source: techiescientist.com
A Lewis electron dot diagram or electron dot diagram or a Lewis diagram or a Lewis structure is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The electrons from one atom are shown as dots and the electrons from the other. The electron transport system refers to the Electron transport chain or ETS in abbreviated form that is present in the inner mitochondrial membraneETS involves electron transfer through a series of protein complexes from higher NADH to lower energy state O 2 by releasing protons into the cytosol. Secondly ET is rather simple and very suitable to be used as a model for studying solvent effects and to relate the kinetics of ET reactions to. This step results in the translocation of four protons similar to complex I across.
 Source: breakingatom.com
Source: breakingatom.com
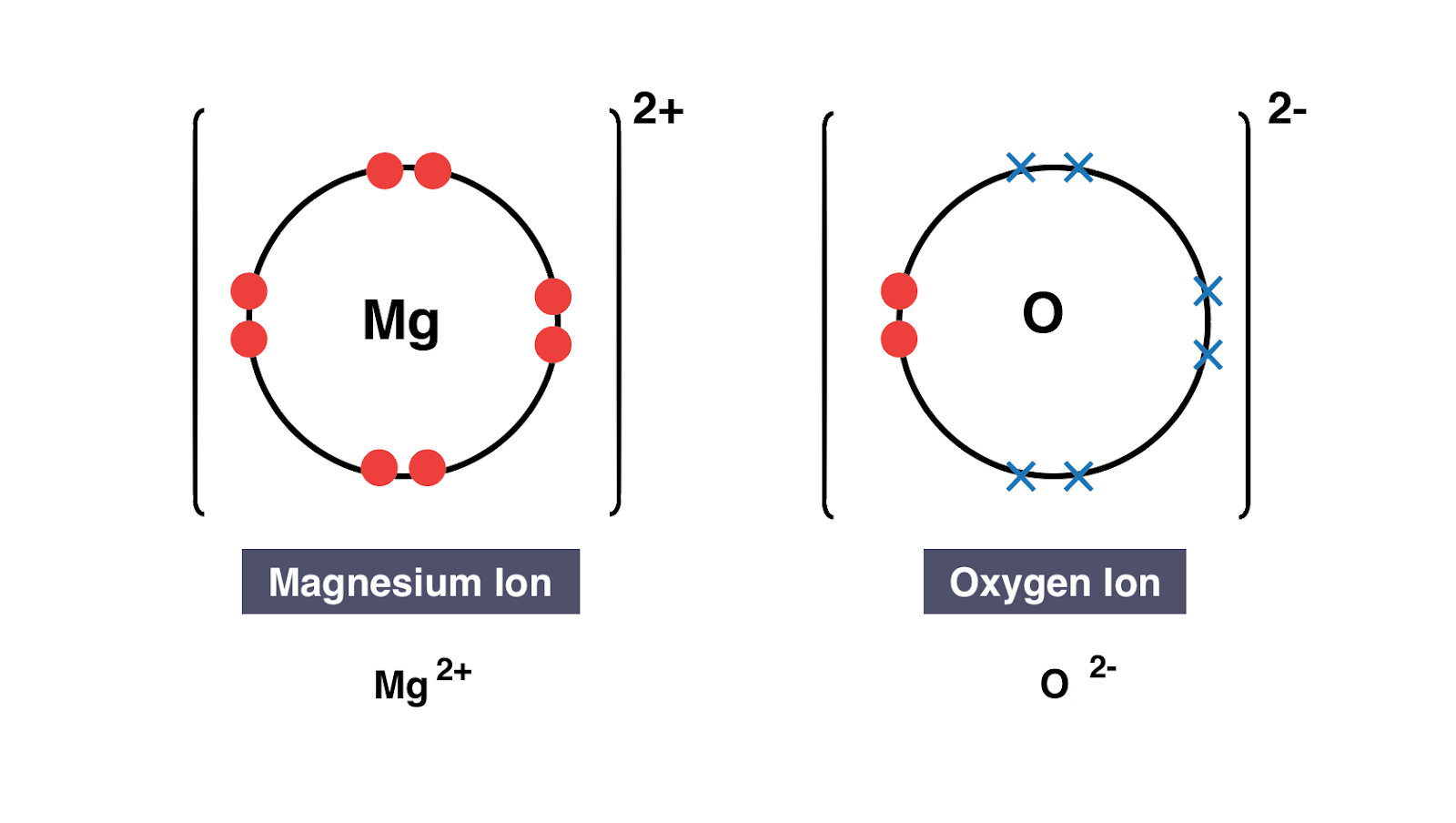
In the transition state of the complex the electrons have equal probability. This leads to the development of an electrochemical proton gradient across the membrane that activates the. A similar process occurs between Mg atoms and O atoms except in this case two electrons are transferred. We show this process with one arrow labeled a in the diagram that starts at a lone pair of electrons on the basic. In electron transfer the number of electrons lost must equal the number of electrons gained.
 Source: igcse-chemistry-2017.blogspot.com
Source: igcse-chemistry-2017.blogspot.com
A Lewis electron dot diagram. ETD is used extensively with polymers and biological molecules such as proteins and peptides for sequence analysis. We saw this in the formation of NaCl. Many of the ions that form have eight electrons in their valence shell. A similar process occurs between Mg atoms and O atoms except in this case two electrons are transferred.
 Source: courses.lumenlearning.com
Source: courses.lumenlearning.com
In Section 91 Lewis Electron Dot Diagrams we saw how ions are formed by losing electrons to make cations or by gaining electrons to form anions. In Section 91 Lewis Electron Dot Diagrams we saw how ions are formed by losing electrons to make cations or by gaining electrons to form anions. Showing the transfer of electrons that takes place between metals and nonmetal atoms - thus forming oppositely charged ions. Complex III Cytochrome bc1 complex. The electron transport chain is made up of a series of spatially separated enzyme complexes that transfer electrons from electron donors to electron receptors via sets of redox reactions.
 Source: igcse-chemistry-2017.blogspot.com
Source: igcse-chemistry-2017.blogspot.com
The astute reader may have noticed something. In both cases the system is represented in two states that before electron transfer R the reactant state and that after electron transfer P the product state. NAD H FMN NAD FMNH 2. In the electron transfer chain electrons move along a series of proteins to generate an expulsion type force to move hydrogen ions or protons across the mitochondrial membrane. Many of the ions that form have eight electrons in their valence shell.
 Source: courses.lumenlearning.com
Source: courses.lumenlearning.com
Showing the transfer of electrons that takes place between metals and nonmetal atoms - thus forming oppositely charged ions. Electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the elements symbol. Paracoccus denitrificans is a gram-negative facultative anaerobic soil. A beryllium atom with two valence electrons would have the electron dot diagram. The example below shows the transfer of a proton from the relatively acidic acetic acid molecule to the relatively basic hydroxide anion.

Showing the transfer of electrons that takes place between metals and nonmetal atoms - thus forming oppositely charged ions. The number of dots equals the number of valence electrons in the atom. ETD is used extensively with polymers and biological molecules such as proteins and peptides for sequence analysis. In this process water splits into hydrogen ions oxygen and both ATP and NADPH 2 are produced. There are about 15 different chemical groups that accept or transfer electrons through the electron chain.
 Source: courses.lumenlearning.com
Source: courses.lumenlearning.com
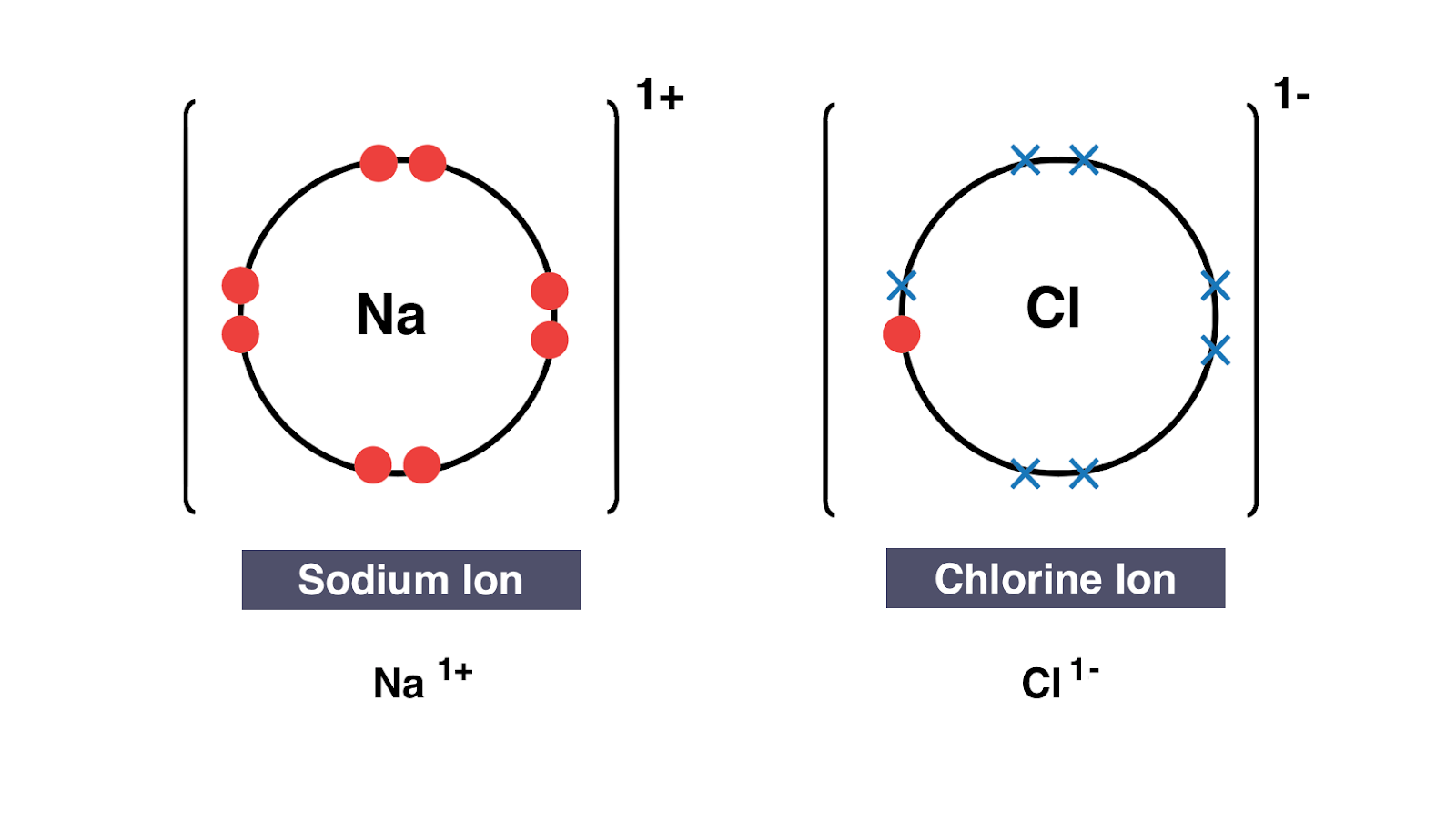
Dot and cross diagrams A dot and cross diagram models the transfer of electrons from metal atoms to non-metal atoms. Many of the ions that form have eight electrons in their valence shell. The number of dots equals the number of valence electrons in the atom. Some bacterial electron transport chains resemble the mitochondrial electron transport chain. A Lewis electron dot diagram or electron dot diagram or a Lewis diagram or a Lewis structure is a representation of the valence electrons of an atom that uses dots around the symbol of the element.
This site is an open community for users to do submittion their favorite wallpapers on the internet, all images or pictures in this website are for personal wallpaper use only, it is stricly prohibited to use this wallpaper for commercial purposes, if you are the author and find this image is shared without your permission, please kindly raise a DMCA report to Us.
If you find this site helpful, please support us by sharing this posts to your own social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title electron transfer diagram by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.